Hardware Solutions
Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Immunology
Dr. Mathieu-Benoît Voisin and Dr. Doris Proebstl, Queen Mary University of London, and colleagues.
When tissue is injured or invaded, leukocytes (also known as white blood cells) migrate through blood vessel walls to reach the site of injury or infection. This complex migration process is a key part of the immune response but is not fully understood. Now, researchers led by Dr. Mathieu-Benoît Voisin and Dr. Doris Proebstl at Queen Mary University of London, have made breakthrough observations of real-time leukocyte migration in vivo thanks to Imaris software, new confocal imaging techniques, and new genetically modified animals.
Leukocyte migration involves complex interactions between immune cells and components of blood vessel walls. Leukocyte transmigration occurs in post capillary venules, which are made of a single layer of cobblestoned endothelial cells and a discontinuous sheath of elongated pericytes that surround the endothelial cells and help to stabilize the vessel. The endothelial cells and the pericytes secrete a network of tightly packed extracellular matrix proteins called the venular basement membrane.
"Our understanding of the interactions between leukocytes and endothelial cells has been considerably enhanced over the past two decades, but only accounts for less than 15 minutes of the total transmigration process,” says Dr. Voisin. “The journey of leukocytes through the blood vessel wall takes 40 to 50 minutes in vivo.”
Imaris allowed the researchers make real-time 3D observations in vivo. They used the Imaris measurement module to analyze intravital confocal images of leukocyte migration in living mice. They measured the fluorescence intensity and morphologic characteristics of cells as well as the localization of molecules. They also tracked 3D cell movement in real time. Since they worked with live animals, the software's ability to correct drift and to compensate for tissue mobility, heartbeat, and blood pressure helped with the analysis and interpretation of data.
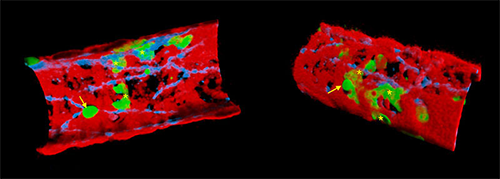
The three cellular players in transmigration: leukocytes (neutrophils in green), the endothelial cells (endothelial cell junctions seen in blue) and the pericytes (in red) are shown as observed by live confocal microscopy. The two images are from a cremasteric venule of a double fluorescent strain of mice with endogenously fluorescent neutrophils (eGFP) and pericytes (RFPCherry) and immunolabeled in vivo for endothelial cell junctions (PECAM-1). The left panel shows the vessel in 3D from the luminal side and exhibiting a rolling neutrophil (arrow) and some neutrophils flattened and trapped between the ECs and the pericytes (stars). On the right, the image is turned 180 degrees to see the vessel from the extravascular space (i.e. tissue). A neutrophil is making its way out of the vessel wall (arrow) whilst others stuck in the pericyte sheath (stars) are looking for the best region to exit the vessel wall. Image courtesy of Dr. Mathieu-Benoît Voisin.
To quantify the leukocyte migration through the pericyte layer, they tracked individual neutrophil migration paths with Imaris. They used these tracks to quantify the duration of transmigration, speed of migration, track length, venular wall track displacement length, and track straightness. They also quantified leukocyte shape by generating an isosurface on neutrophils at different stages of migration.
The imaging analysis revealed close interactions between the cells, such as leukocytes moving along pericytes processes. The researchers could see how the immune cells eventually crossed the blood vessel wall before reaching the tissue. Overall, the study showed several ways in which pericytes support leukocyte migration through vessel walls.
A better understanding of the mechanisms involved in this process could allow the cells to be manipulated in a way that promotes or inhibits leukocyte recruitment. This, in turn, could lead to the development of new potential molecular targets to control inflammatory responses. The researchers will next investigate if and how other leukocytes penetrate the pericyte layer of vessel walls and will examine the molecular mechanisms involved during models of pathological diseases.
Research Paper: Proebstl D, Voisin MB, Woodfin A, Whiteford J, D'Acquisto F, Jones GE, Rowe D, Nourshargh S. Pericytes support neutrophil subendothelial cell crawling and breaching of venular walls in vivo.J Exp Med. 2012 Jun 4;209(6):1219-34. Epub 2012 May 21.
